 To kick-start β-casein degradation by the L. lactis and to discover (putative) bioactive peptides obtained upon degradation of β-casein by the organism. The aim of this study was to build an analytical framework for the analysis of the intracellular peptidome of L. SEARCHGUI ENZYME DEFINITION FULLOn the other hand, only utilizing the culture supernatant of proteolytically active cells does not exploit the full potential of the LAB, as in that case only the proteinase specificity is being utilized while the activities of the more-than-10 intracellular peptidases and possible hidden intracellular bioactive peptides are being ignored. From an application point of view, the costs of employing purified enzymes are too high for industrial-scale use. These studies have identified numerous casein-derived bioactive peptides, most of which having ACE-inhibitory activity. Second, an LAB cell culture is mixed with milk proteins, and the supernatant is subsequently further characterized ( 9 – 11). First, casein proteins are either digested by a purified digestive enzyme (trypsin) or LAB proteinase(s), after which the obtained products are identified ( 6 – 8). Two major ways of bioactive-peptide discovery can be discerned. In the third and last step, multiple peptidases hydrolyze the oligopeptides into smaller peptides and, ultimately, into amino acids that are then available for de novo protein synthesis and other metabolic activities ( 2).Ī great deal of research has focused on the production of milk-derived bioactive peptides using LAB. Second, (a selection of) these peptides are internalized via an oligopeptide transport system (Opp). First, the extracellular cell envelope-associated proteinase (PrtP) degrades casein into oligopeptides. Casein degradation is accomplished in a three-step process. Lactococcus lactis, for instance, depends on the milk protein casein as the major source of nitrogen, since it is auxotrophic for several amino acids. The potential of LAB as production organisms for functional foods is still largely unexplored. However, the value of those fermented products, such as yogurt or cheese, has remained restricted to providing basic nutrition, flavor, and texture. LAB were used as starter cultures for dairy production more than 100 years ago, which has given them their current industrial and economic importance ( 1). Members of the diverse group of lactic acid bacteria (LAB) have been associated with food fermentations since ancient times. This comprehensive pipeline offers a model for discovery of bioactive peptides in combination with other proteins and might be applicable to other bacteria. 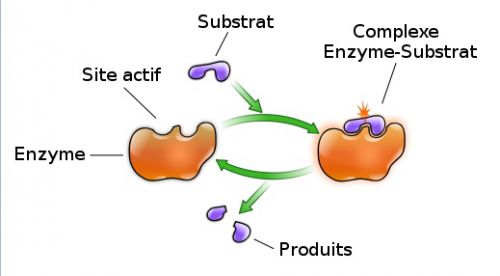
The results suggest that both the number of different bioactive peptides and the bioactivity diversity can be increased by editing the proteolytic system of L. 
We observed a clearly strain-dependent accumulation of peptides with several bioactivities, such as angiotensin-converting enzyme (ACE)-inhibitory, dipeptidyl peptidase 4 (DPP-IV)-inhibitory, and immunoregulatory functions. lactis MG1363 and a set of 6 isogenic multiple peptidase mutants incubated with β-casein. We used the pipeline to examine cultures of L. In addition, we proofread the publicly available bioactive-peptide databases and obtained an optimized database of bioactive peptides derivable from bovine β-casein. lactis, used as an “enzyme complex” to digest β-casein, an extraction method for its intracellular peptidome, and a peptidomics data analysis and visualization procedure. 
It entails an optimized sample preparation protocol for L. Here, we present an analytical pipeline for large-scale intracellular peptidomics of Lactococcus lactis. Large-scale mass spectrometry-based peptidomics for bioactive-peptide discovery is relatively unexplored because of challenges in intracellular peptide extraction and small-peptide identification. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
